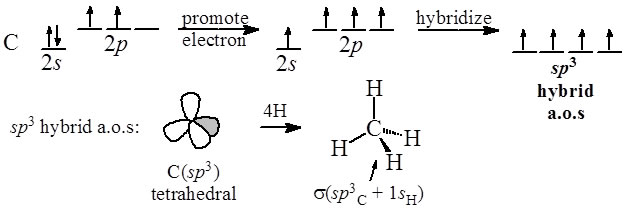
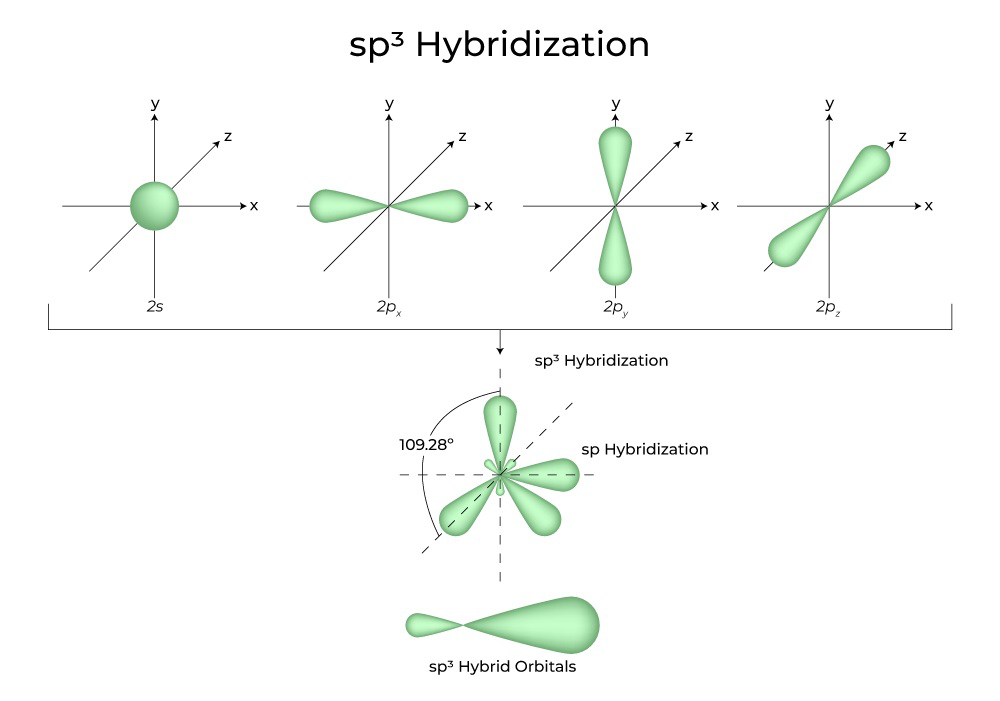
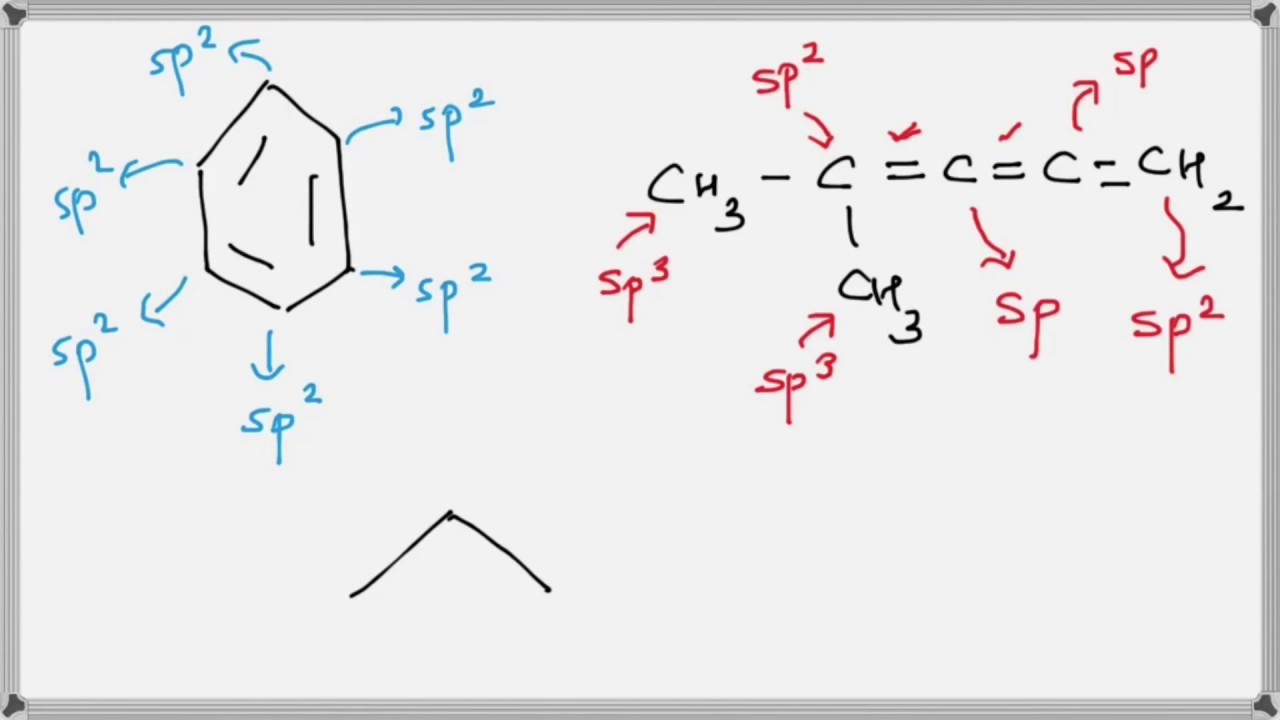
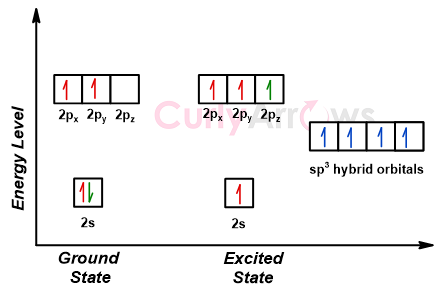
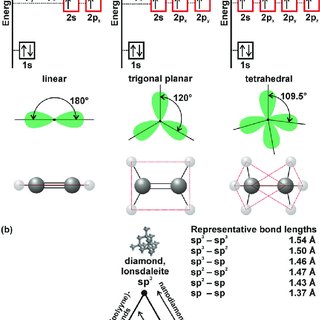
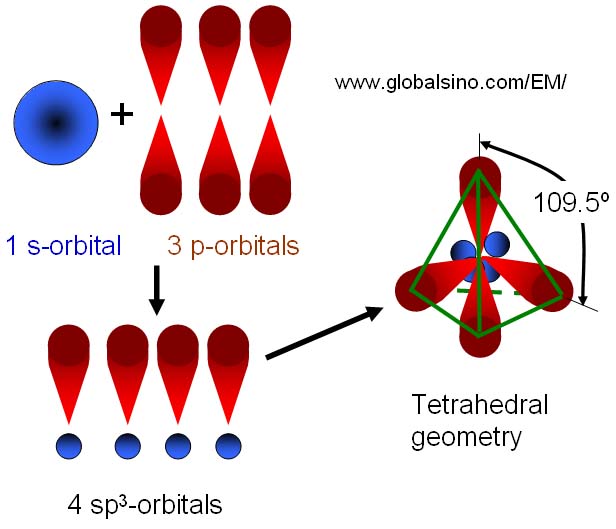
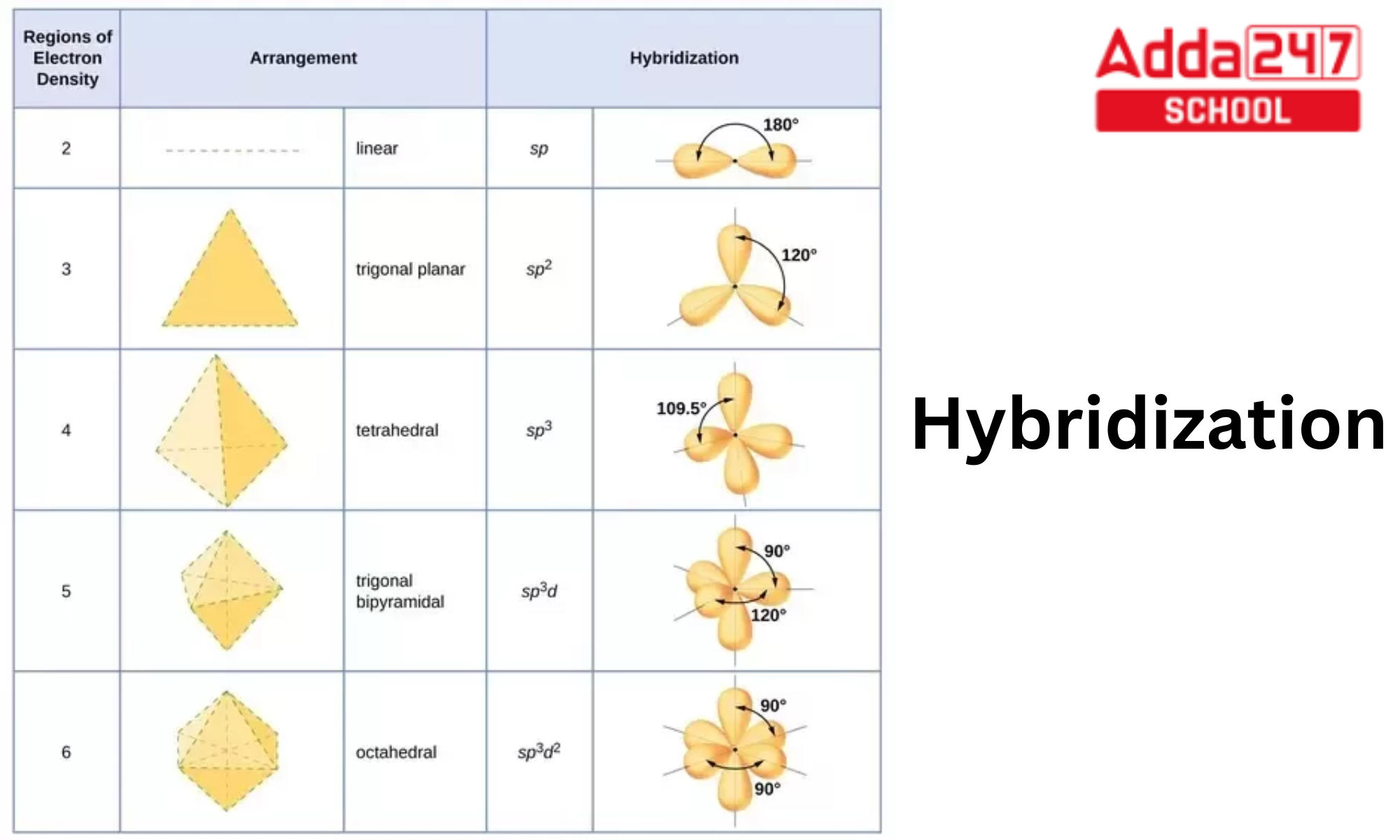
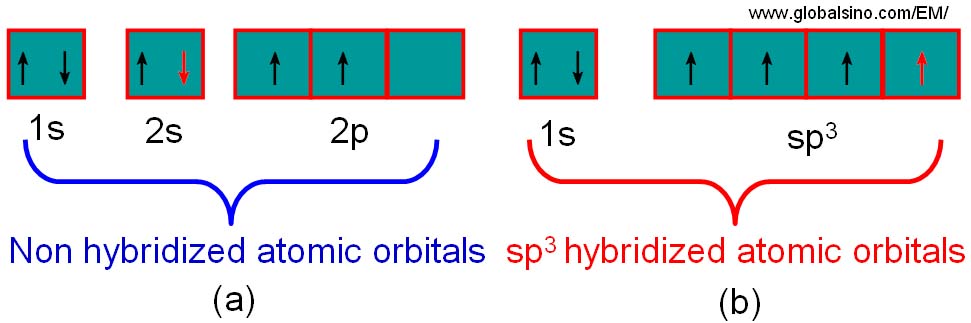
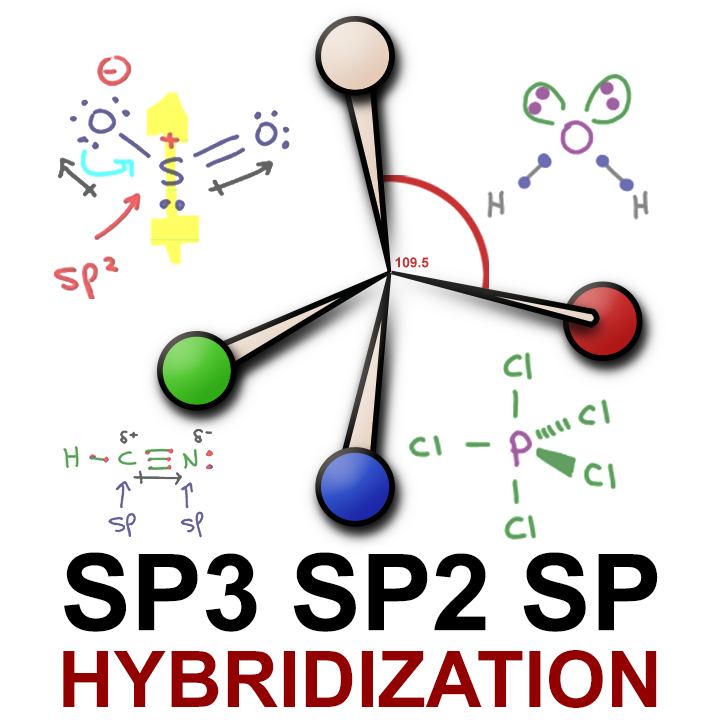
What is the geometry of: (a) an sp3 hybridized carbon atom? (b) an sp2 hybridized carbon atom? (c) an sp hybridized carbon atom? (d) a trivalent nitrogen atom (three bonds on the

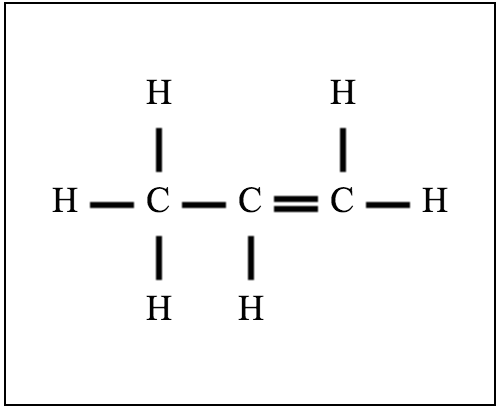
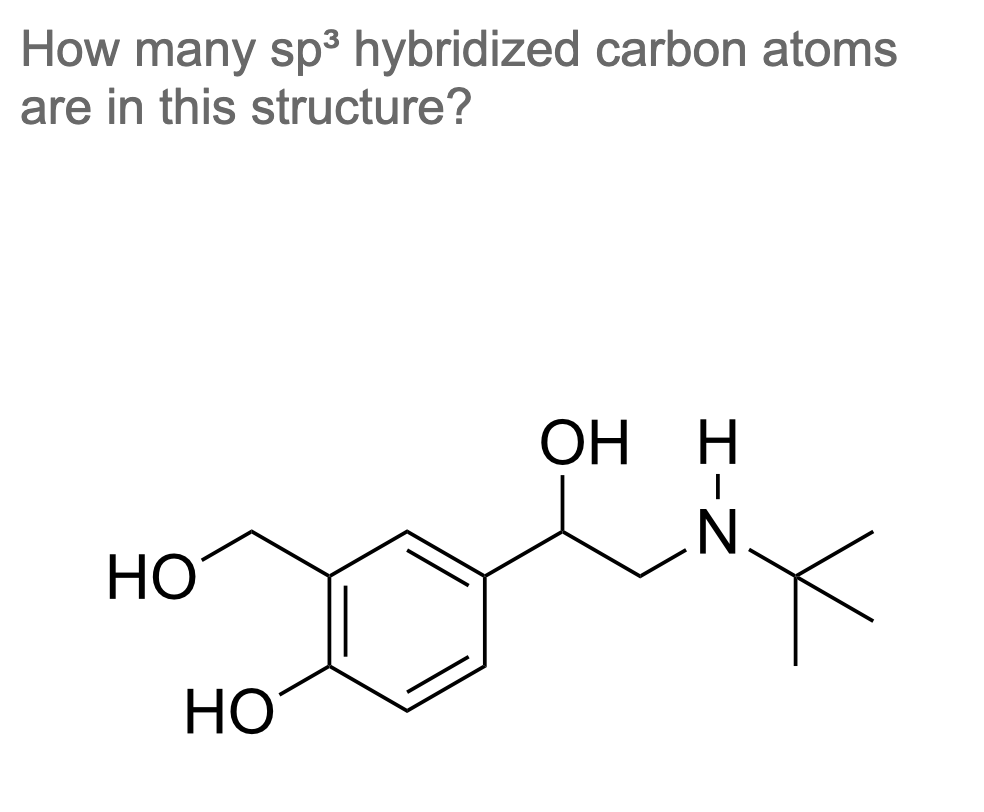
A structure for a molecule that meet the following description: Contains two sp2-hybridized carbons and two sp3-hybridized carbons | Homework.Study.com