
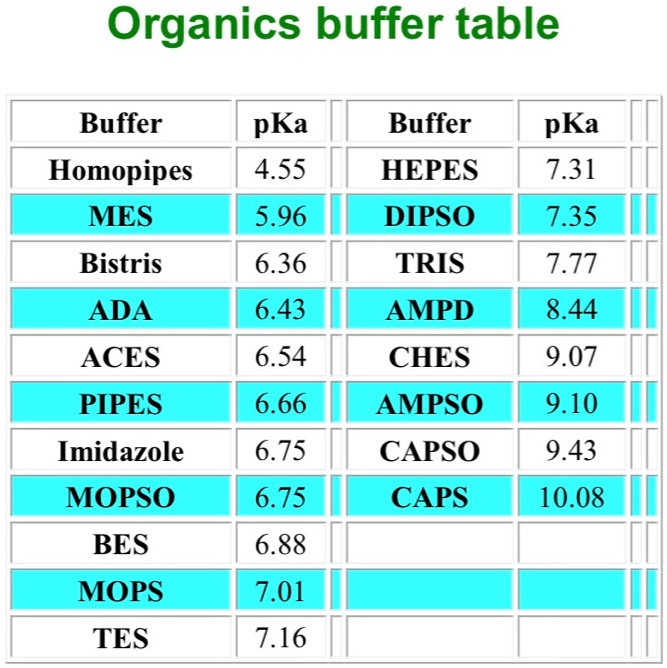
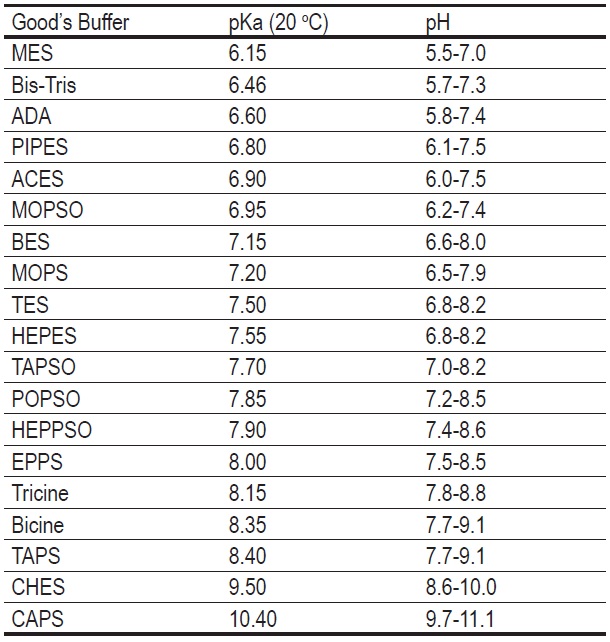
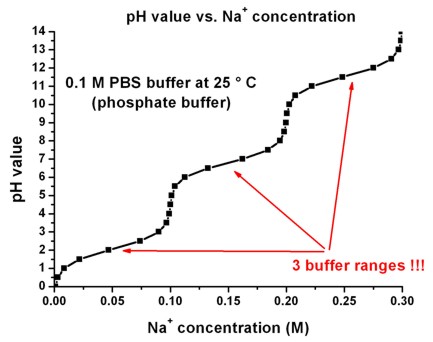
Dissociation steps and pKa values at 25 °C and 37 °C of the buffers... | Download Scientific Diagram
Analysis of Good's buffer species MES and MOPS. (a) Chemical structure... | Download Scientific Diagram

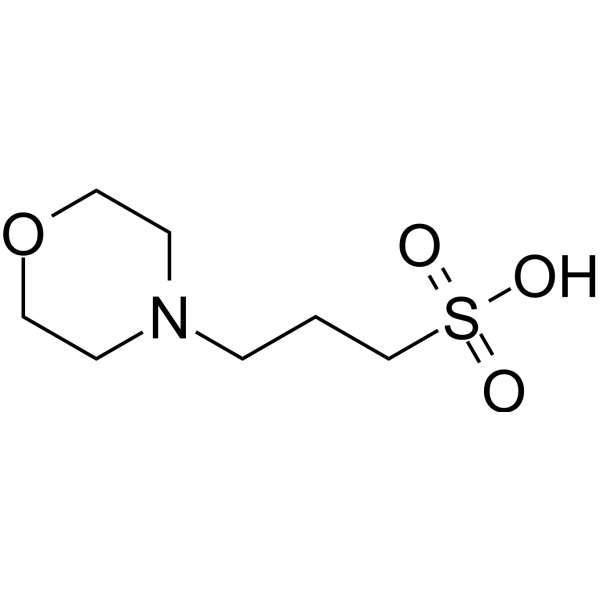
MOPS, Free Acid, Ultrol Grade - CAS 1132-61-2 - Calbiochem A zwitterionic buffer useful in the pH range of 6.5-7.9. Has a pKa of 7.20 at 25 C. Absorbance (1.0 M, H2O, 260 nm): 0.05. 1132-61-2
![MOPS Sodium Salt [3-(N-Morpholino)-propane sulfonic acid sodium salt] - 71119-22-7 - Discovery Fine Chemicals MOPS Sodium Salt [3-(N-Morpholino)-propane sulfonic acid sodium salt] - 71119-22-7 - Discovery Fine Chemicals](https://discofinechem.stonespiral-staging.co.uk/wp-content/uploads/2015/08/71119-22-7.png)
MOPS Sodium Salt [3-(N-Morpholino)-propane sulfonic acid sodium salt] - 71119-22-7 - Discovery Fine Chemicals

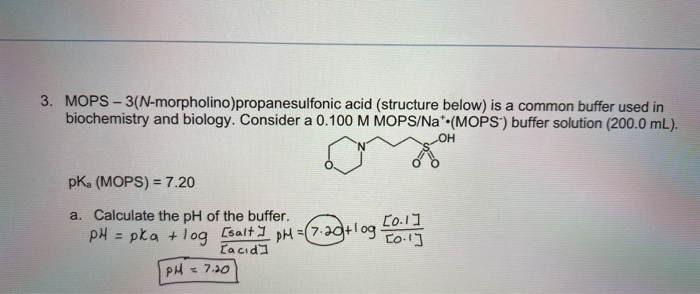
You prepare a buffer solution from 10.0 mL of 0.100 M MOPS (3- morpholinopropane-1-sulfonic acid) and 10.0 mL of 0.077 M NaOH. Next, you add 1.00 mL of 2.05 x 10^-5 M
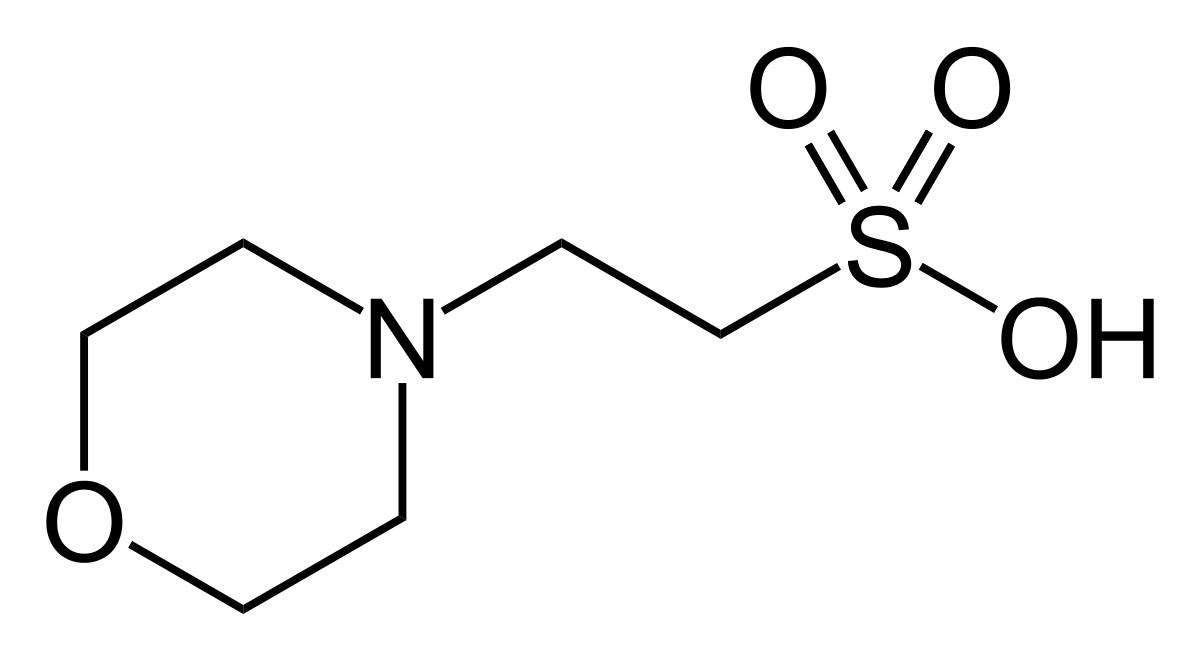
![3[N-Morpholino]propane sulfonic acid | C7H15NO4S - PubChem 3[N-Morpholino]propane sulfonic acid | C7H15NO4S - PubChem](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=70807&t=l)


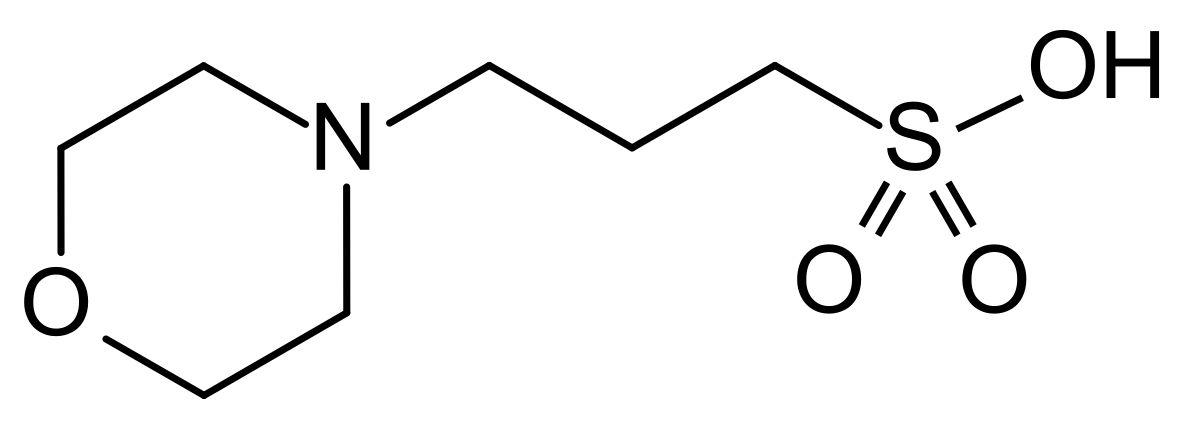







![MOPS - Ácido 3-[N-Morfolino] propanosulfonico • Êxodo Cientifica MOPS - Ácido 3-[N-Morfolino] propanosulfonico • Êxodo Cientifica](https://exodocientifica.com.br/wp-content/uploads/2020/07/MOPS.jpg)
![Solved 7) MOPS (3-[N-morpholino]propanesulfonic) acid can be | Chegg.com Solved 7) MOPS (3-[N-morpholino]propanesulfonic) acid can be | Chegg.com](https://media.cheggcdn.com/study/3f1/3f1d2253-902e-4765-805c-d25932f0e9d3/image)