![PDF] Effects of CO2 on H2O band profiles and band strengths in mixed H2O:CO2 ices | Semantic Scholar PDF] Effects of CO2 on H2O band profiles and band strengths in mixed H2O:CO2 ices | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/d35b84dc8965905b09b78fee990836ee8b77fcd6/6-Table5-1.png)
PDF] Effects of CO2 on H2O band profiles and band strengths in mixed H2O:CO2 ices | Semantic Scholar
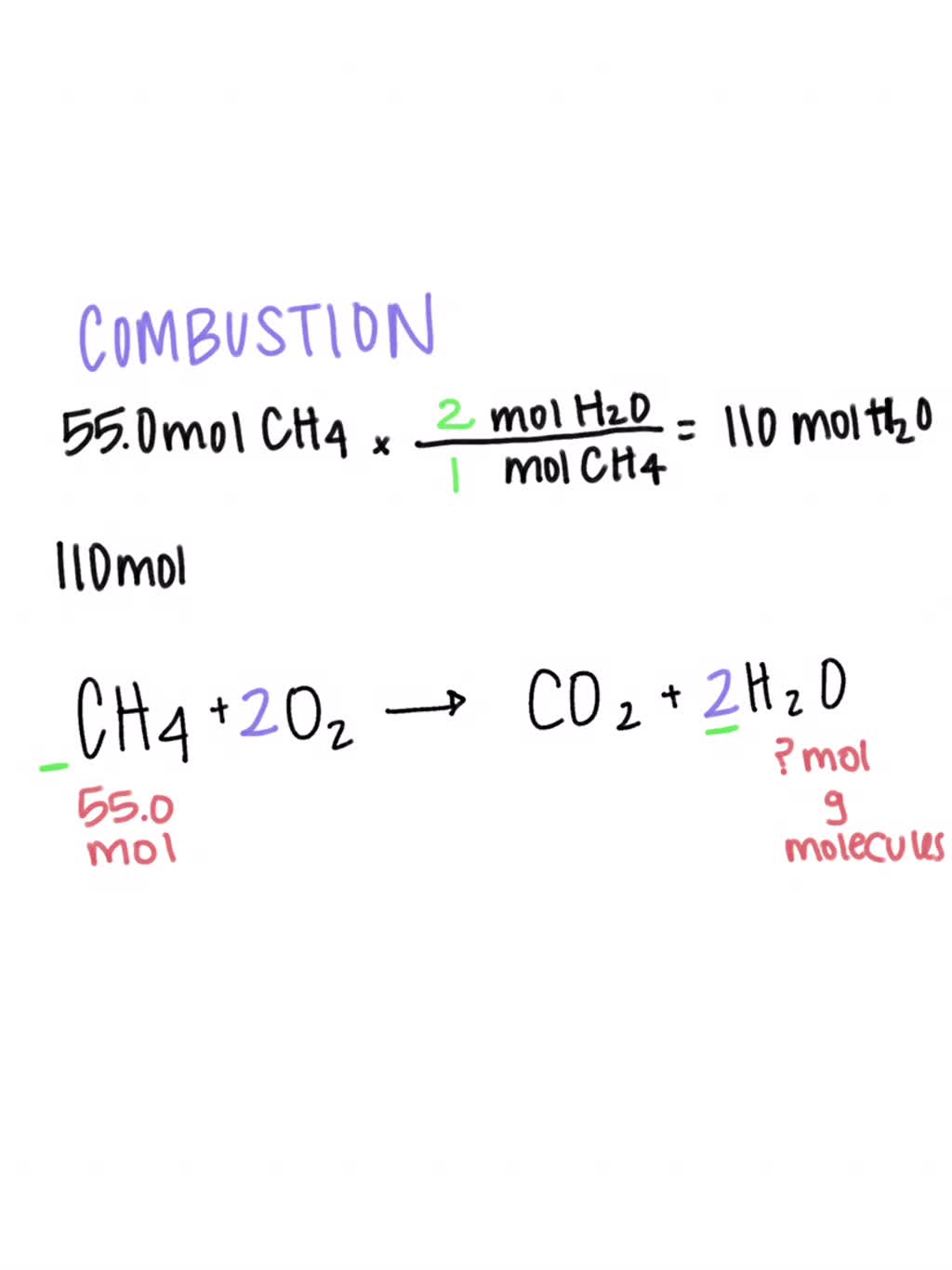
how to balance chemical equation c6h6+o2 arrow mark co2+h20 but. i tried and in RHS why should we put 12 next to carbon instead of that we can put put 3 so
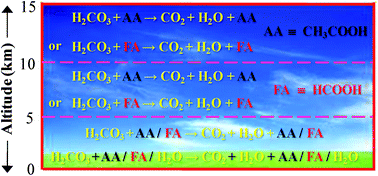
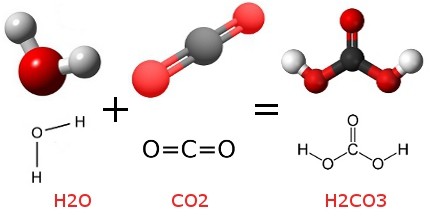
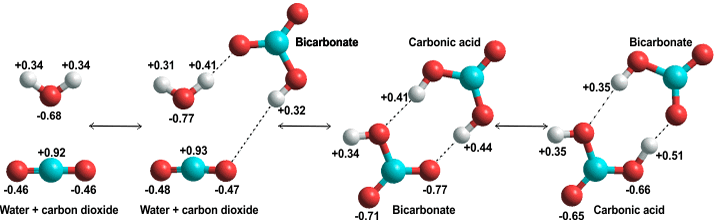
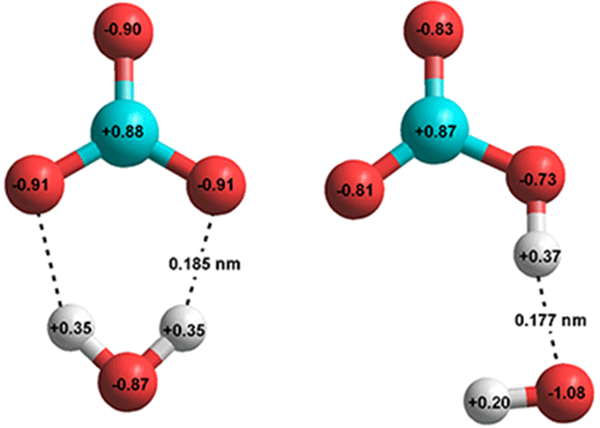
H2CO3 → CO2 + H2O decomposition in the presence of H2O, HCOOH, CH3COOH, H2SO4 and HO2 radical: instability of the gas-phase H2CO3 molecule in the troposphere and lower stratosphere - RSC Advances (
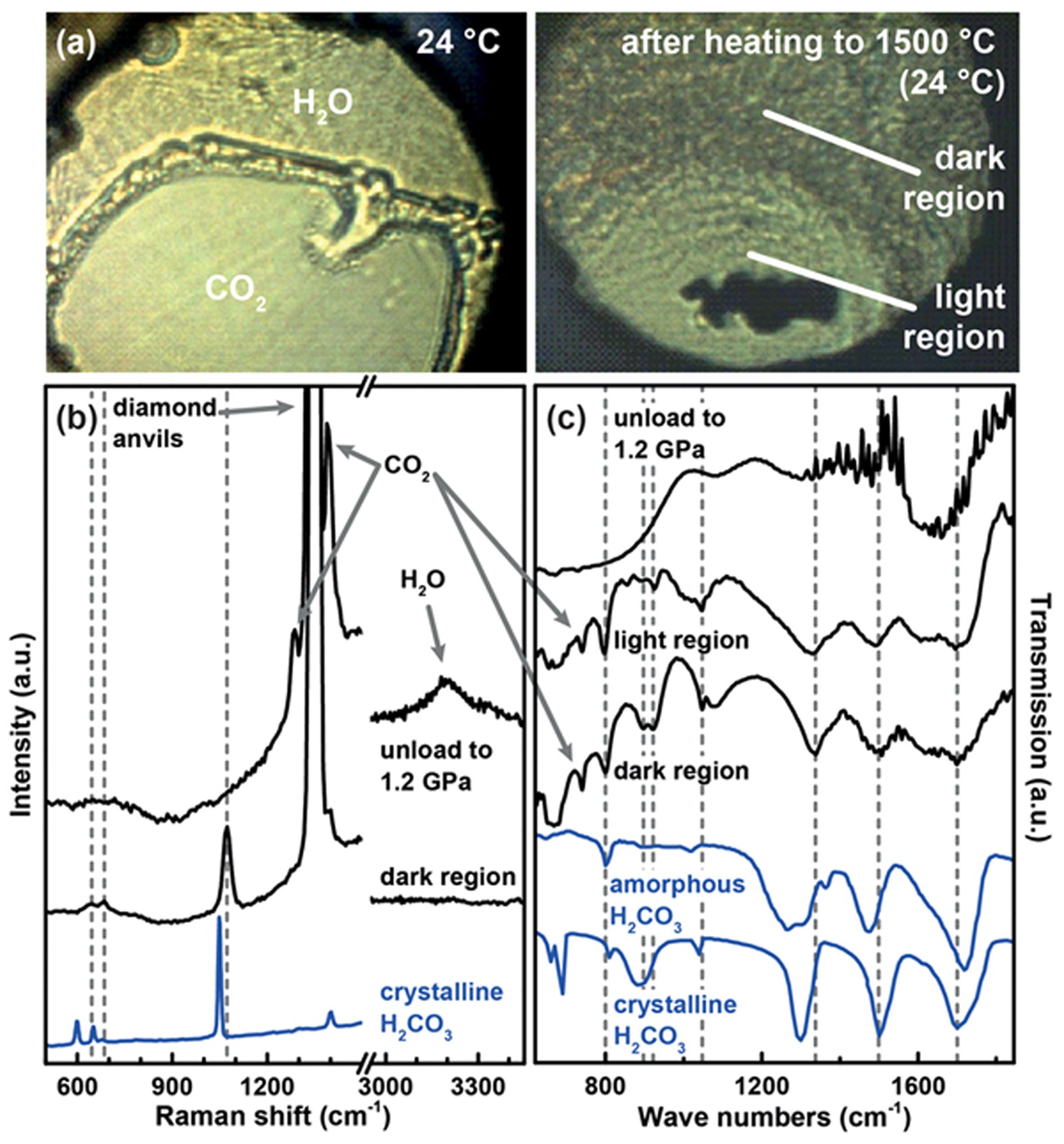
Stable solid and aqueous H2CO3 from CO2 and H2O at high pressure and high temperature | Scientific Reports

The promoting effects of CO2 and H2O on selective hydrogenations in CO2/H2O biphasic system - ScienceDirect
Write fully balanced equations for the following : (a) CO2 + H2O → ............ - Sarthaks eConnect | Largest Online Education Community
![Identify Bronsted - Lowry acids in the given reaction. [ Al (H2O)6 ]^3 + + H CO3^- [ Al (H2O)5 (OH^-) ]^2 + + H2CO3 A B C D Identify Bronsted - Lowry acids in the given reaction. [ Al (H2O)6 ]^3 + + H CO3^- [ Al (H2O)5 (OH^-) ]^2 + + H2CO3 A B C D](https://dwes9vv9u0550.cloudfront.net/images/4940114/ca6cb326-72a5-4e75-8a33-8b87ccbb46e4.jpg)
Identify Bronsted - Lowry acids in the given reaction. [ Al (H2O)6 ]^3 + + H CO3^- [ Al (H2O)5 (OH^-) ]^2 + + H2CO3 A B C D