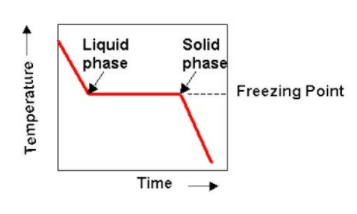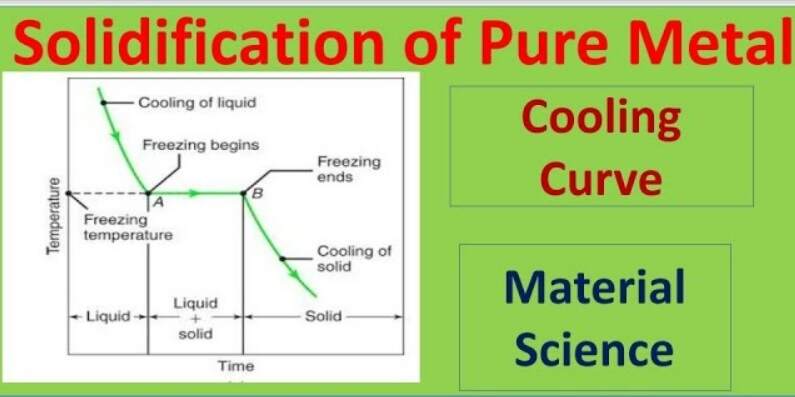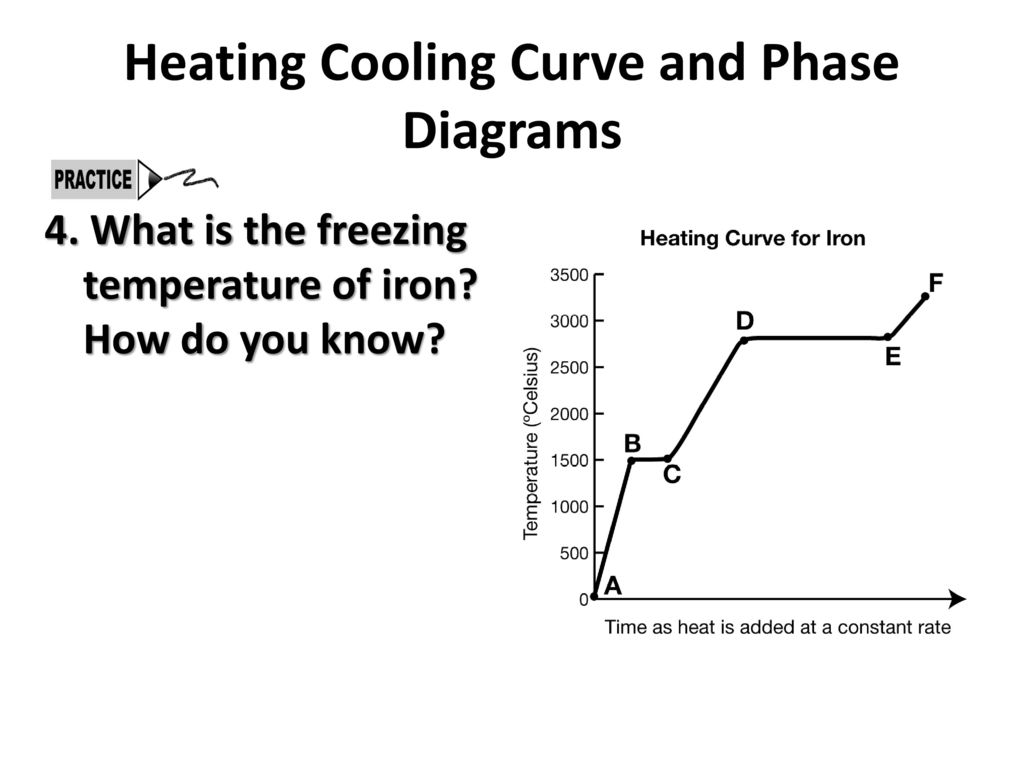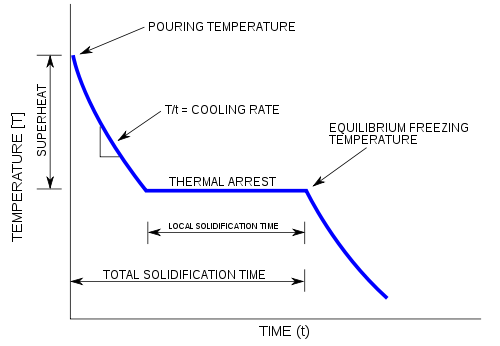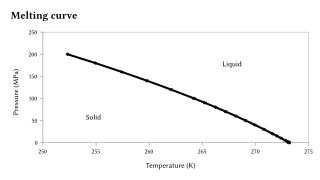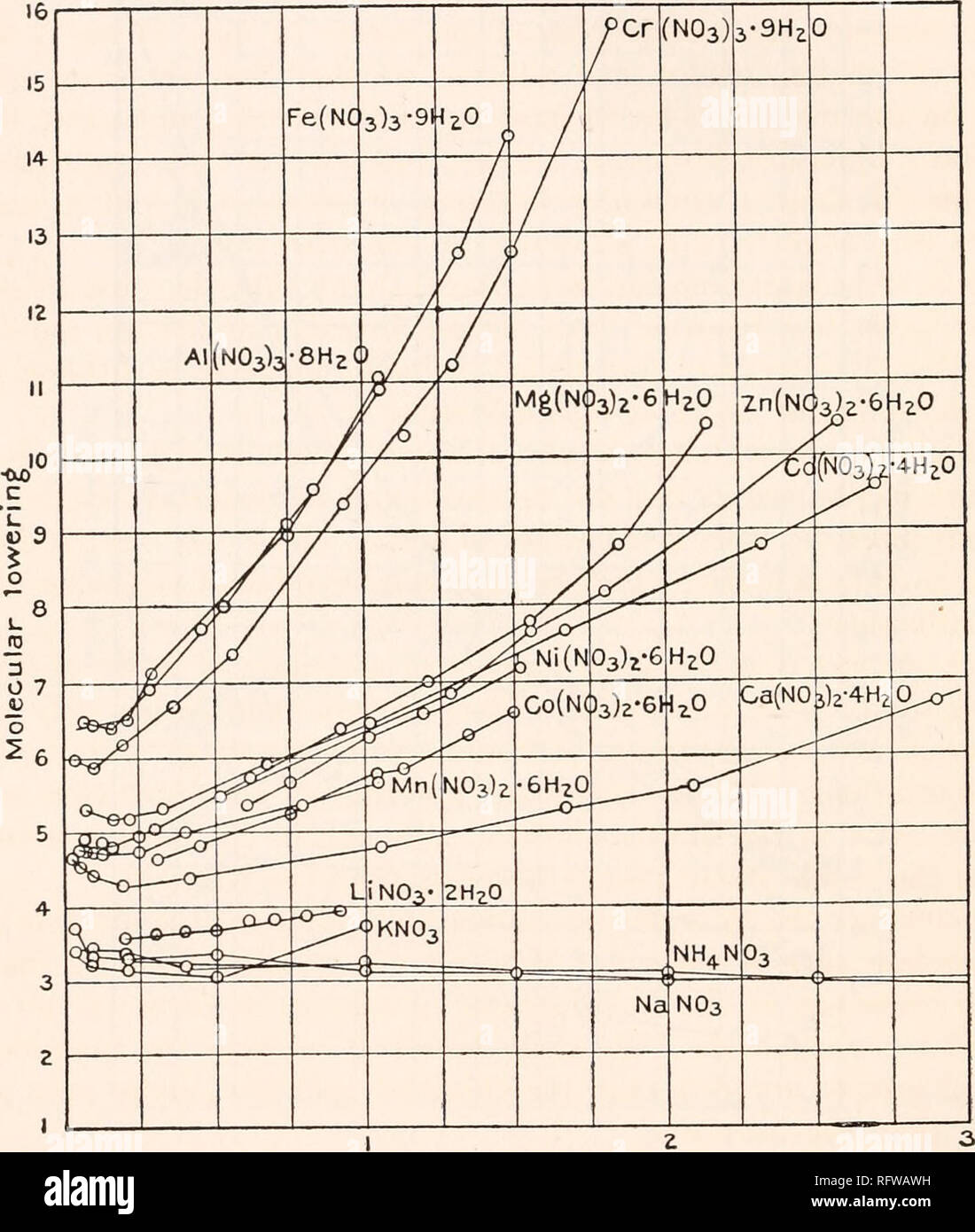
Carnegie Institution of Washington publication. 24 HYDRATES IN AQUEOUS SOLUTION. with this substance, to see if some error had not found its way into this part of the work, and the
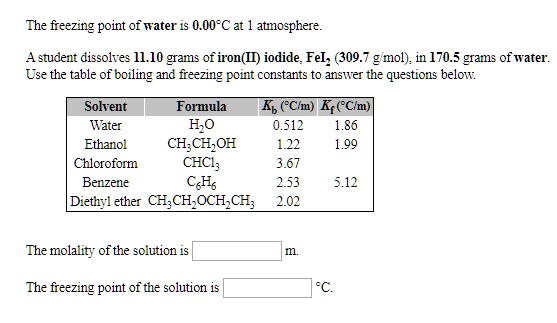
SOLVED: The freezing point of water 0.00-C at atmosphere: Astudent dissolves Il.1O grams of iron(ID) iodide; Fel; (309.7 g mol) in 170.3 grams of water: Use the table of boiling and freezing
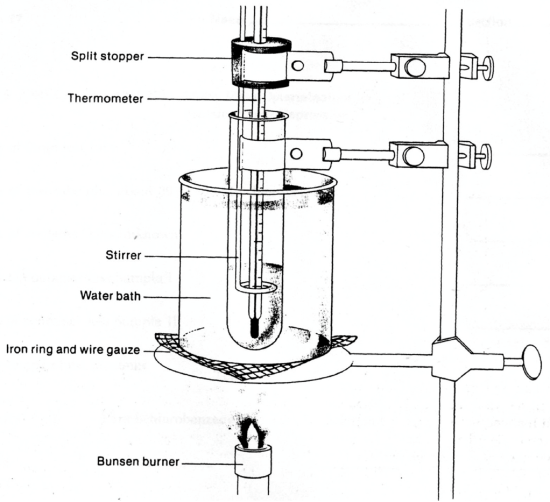
10: Determination of the Molar Mass by Freezing Point Depression (Experiment) - Chemistry LibreTexts

Freezing temperatures of aqueous iron(III) sulfate solutions and crystallization of a new acidic water-rich sulfate - ScienceDirect

Basic ice-modulation functions of CPAs. Ice freezing point depression,... | Download Scientific Diagram

📈a scientist has a mystery element that she wants to identify by using its properties these - Brainly.com

Boiling point boiling point: the temperature at which a substance changes from a liquid to a gas The BOILING POINT of pure water is 100° Celsius. - ppt download

Freezing temperatures of aqueous iron(III) sulfate solutions and crystallization of a new acidic water-rich sulfate - ScienceDirect