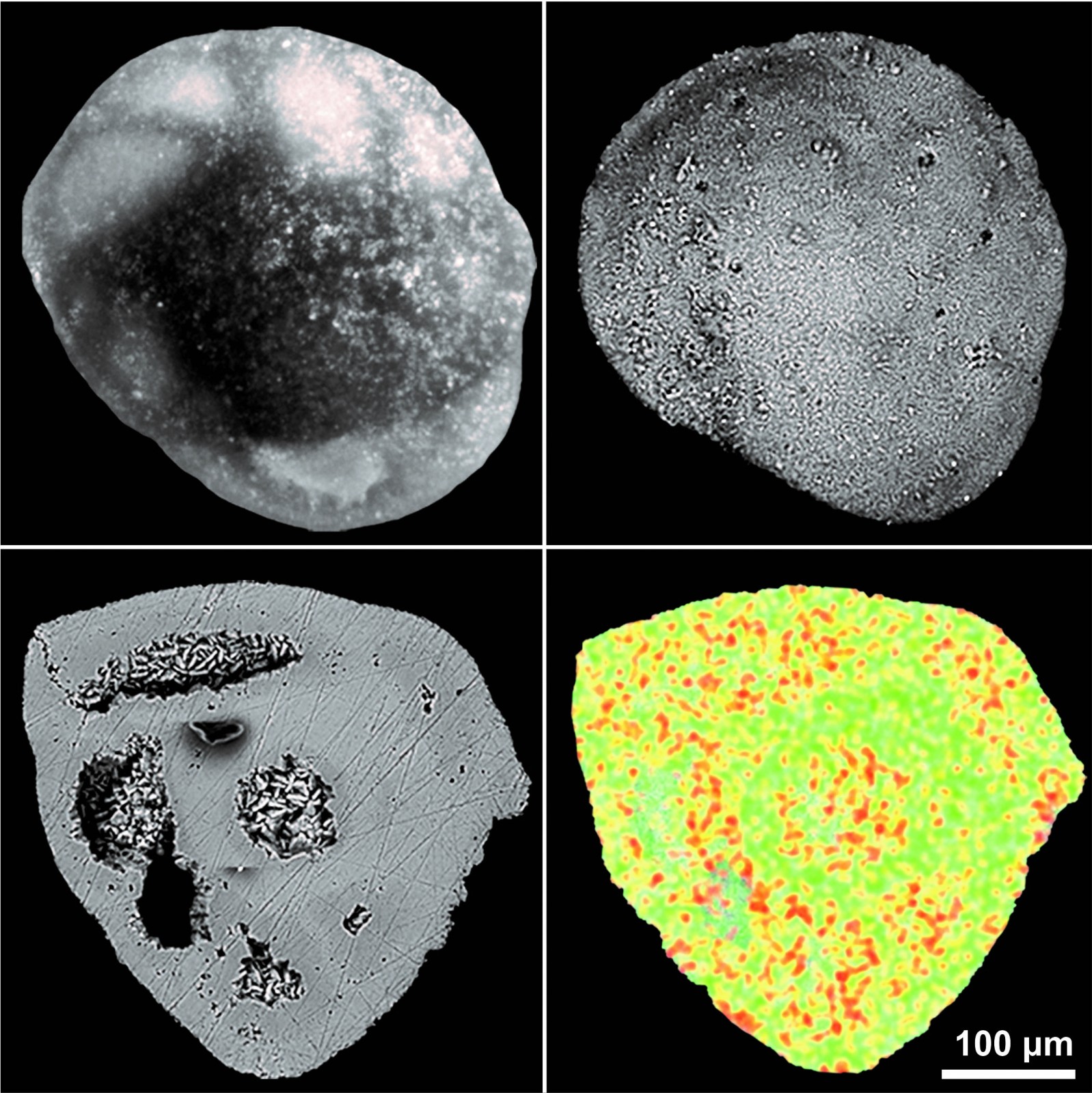
Fast evaluation of inhibitors for calcium carbonate scale based on pH continuous measurements in jar test at high salinity condition - ScienceDirect

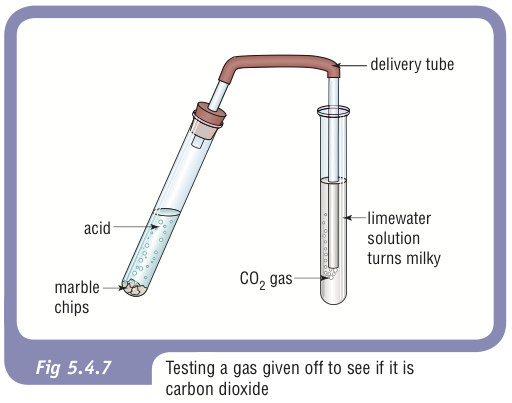
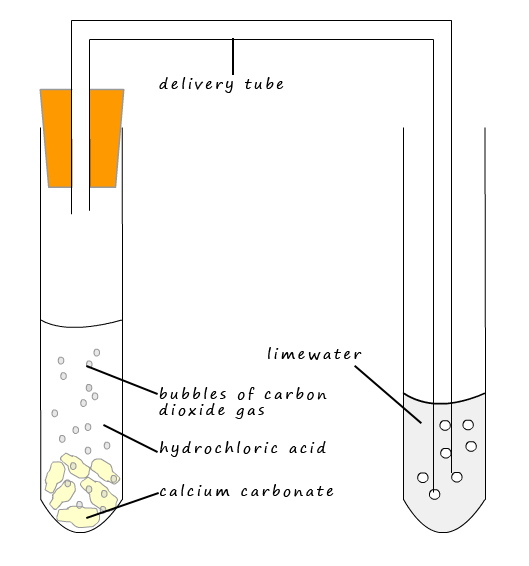
Take some calcium carbonate (marble pieces) in a boiling tube as shown in figure. Add dilute hydro-chloric acid to it through a thistle funnel. Pass the evolving gas through lime water taken

Limestone: Calcium Carbonate (CaCO3) - Uses, Preparation, Properties, Formula & Structure of Calcium Carbonate

Describe an experiment to study the speed of reaction between calcium carbonate and dilute hydrochloric acid, by measuring the loss in mass of reaction system over time. - Study notes, tips, worksheets,

Diagram for the testing of carbon dioxide gas. bubble carbondioxide through limewater, it forms a solid precipitate of calcium carbonate and lime water turns milky. 30186789 Vector Art at Vecteezy